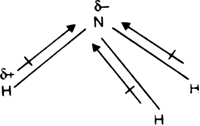
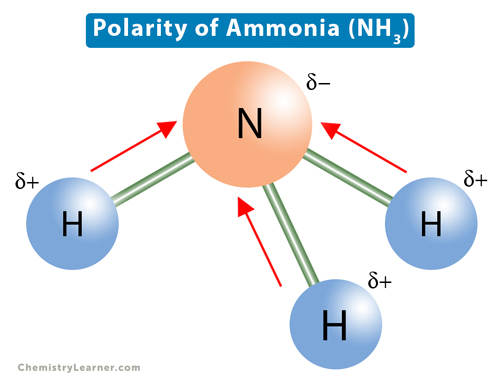
What is the correct dipole moment of $N{{H}_{3}}$ and $N{{F}_{3}}$ respectively?(A)- $4.90\\times {{10}^{-30}}$ C m and $0.80\\times {{10}^{-30}}$ C m(B)- $0.80\\times {{10}^{-30}}$ C m and $4.90\\times {{10}^{-30}}$ C m(C)- $4.90\\times {{10}^{-30}}$ C

Is NH3 polar Or Nonpolar? - nh3 Intermolecular Forces, nh3 charge, nh3 bond angle, detailed facts? - chemwhite.com

Can you explain why NH3 has such a large dipole moment compared with NF3? Show work. | Homework.Study.com

Effect of on dipole moments of CO, O3, NH3 molecules Dipole moment |P e... | Download Scientific Diagram

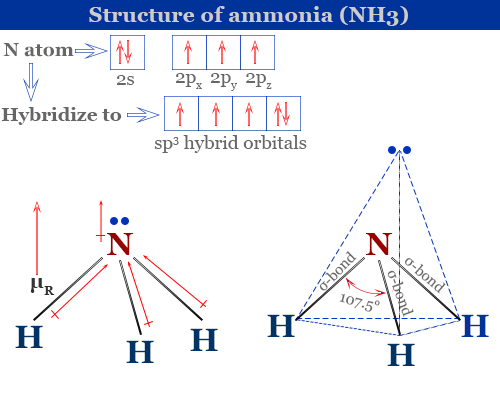




![Which out of \\[N{H_3}\\] and $N{F_3}$ has a higher dipole moment and why? Which out of \\[N{H_3}\\] and $N{F_3}$ has a higher dipole moment and why?](https://www.vedantu.com/question-sets/a32afb56-fc67-43ca-8a5c-e06f6fe2b15f5994708008049533229.png)