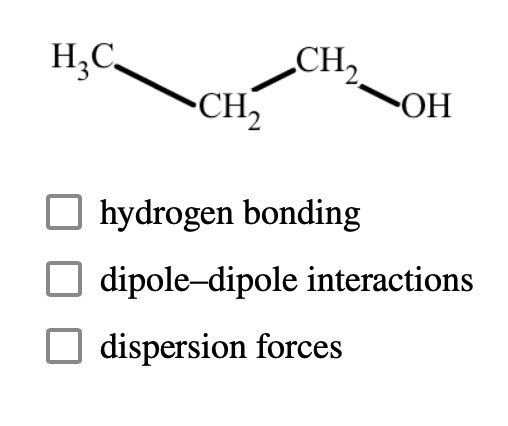
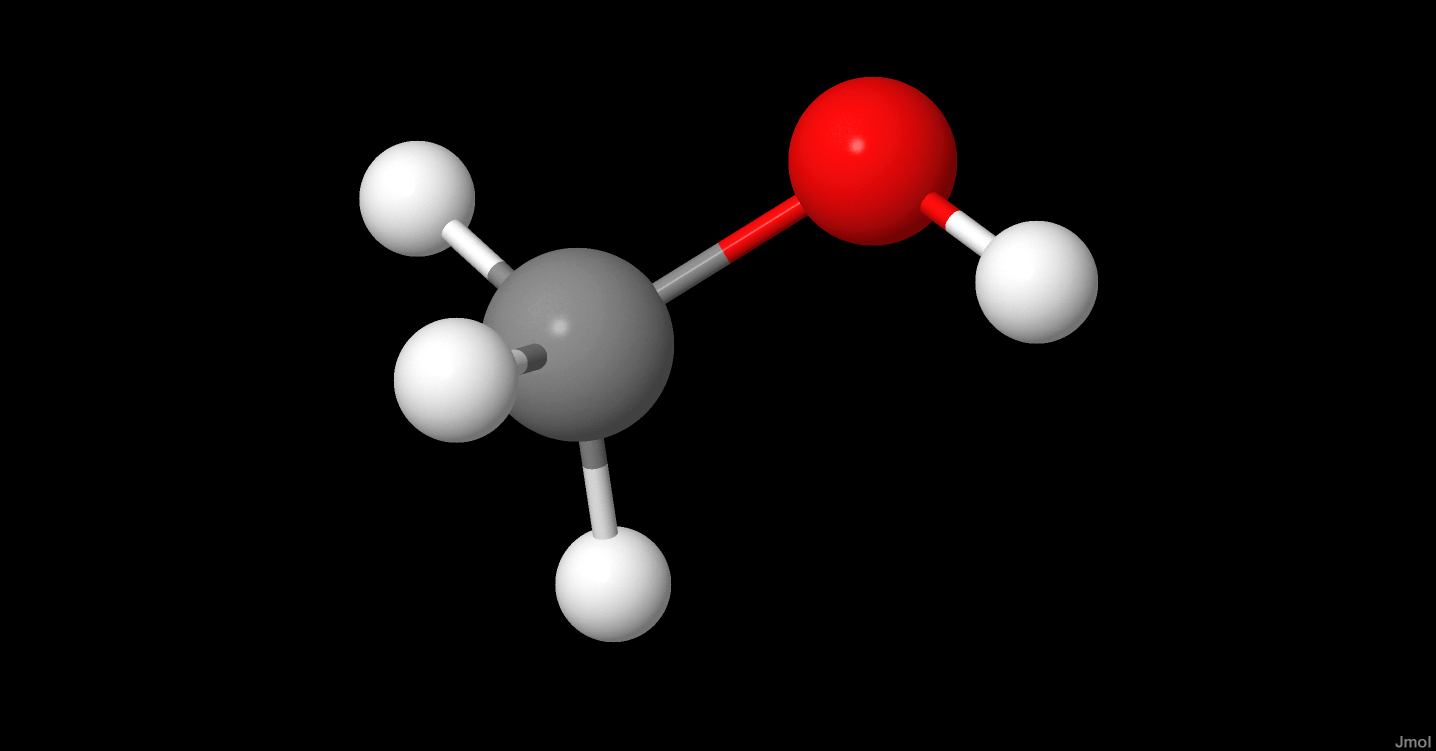
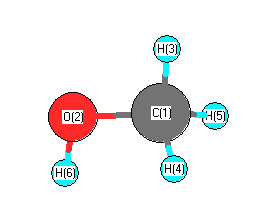
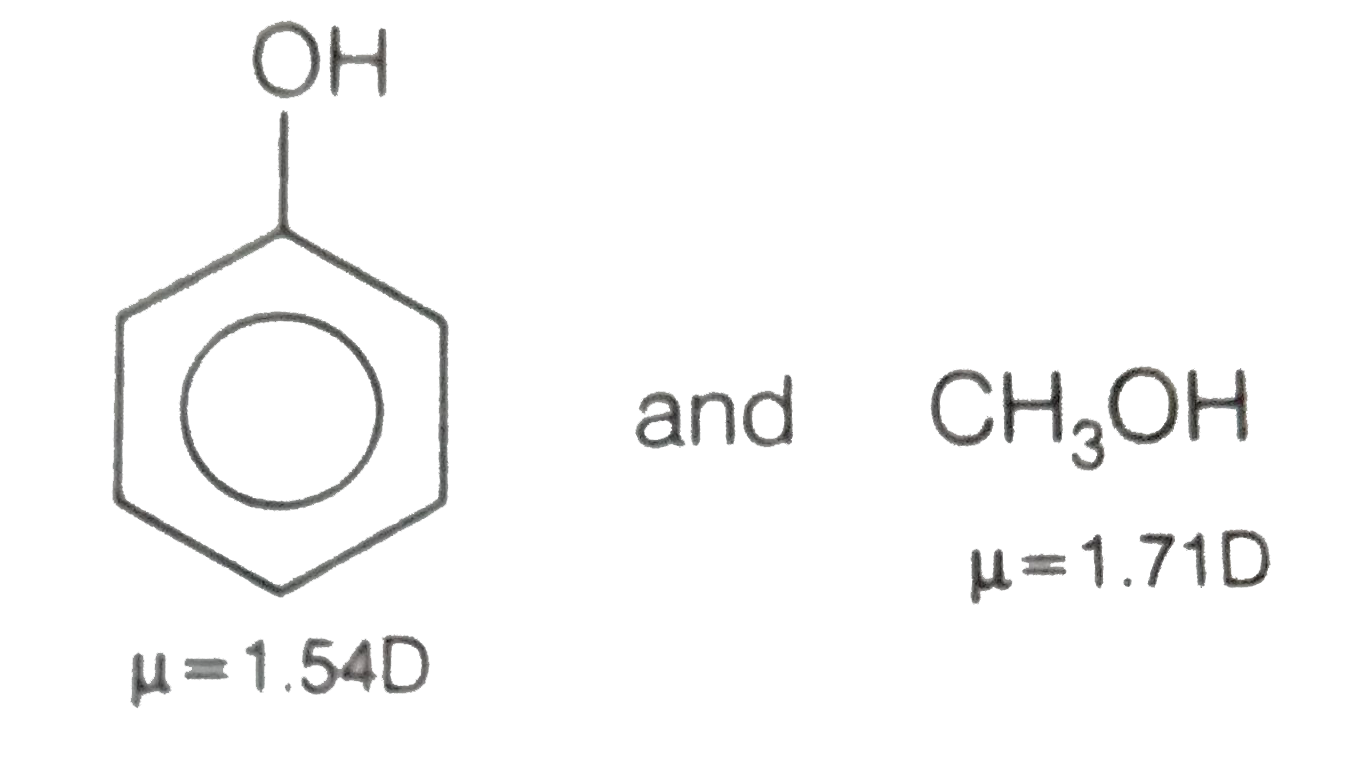
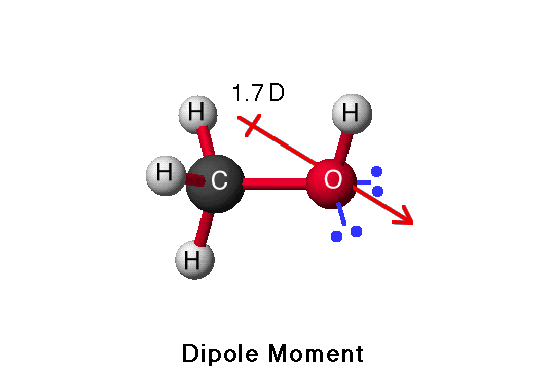
SOLVED:The dipole moment of methanol is μ=1.70 D. Use arrows to indicate the direction in which electrons are displaced.

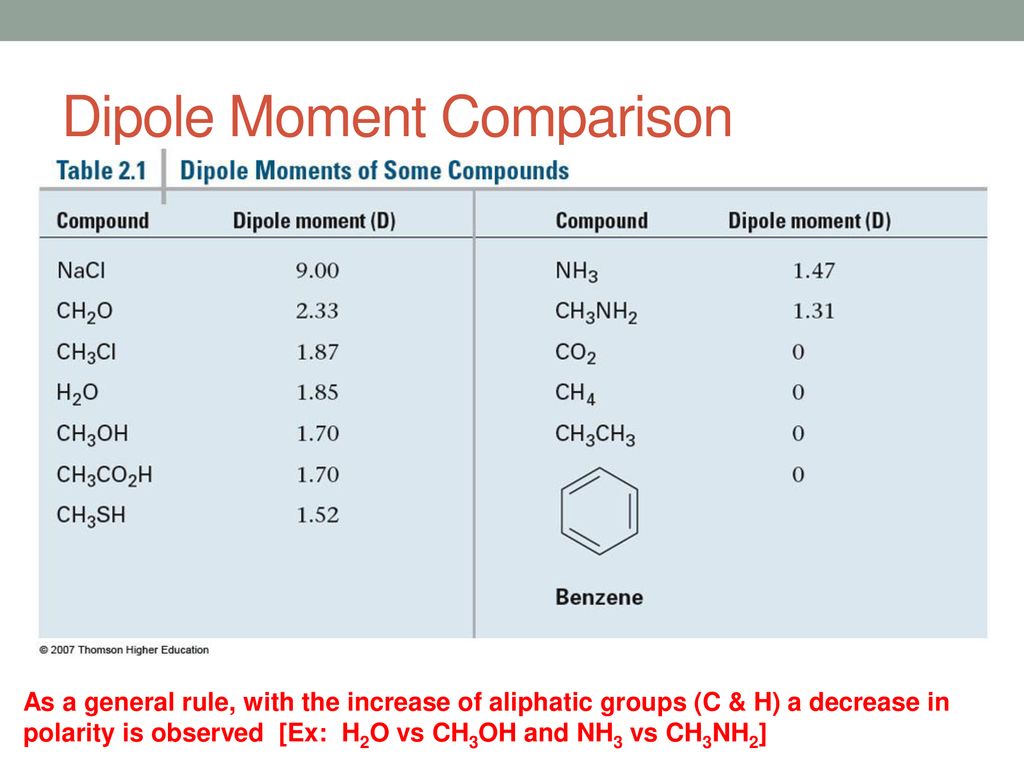
Chemistry Conceptions - Dipole Moments of some compounds. Note: Benzene, CO2 and CH4 has zero dipole moment #SHAREIT. Photo Credit: Chemistry Concepts | Facebook

20. Arrange the following in the order given below: a) Decreasing dipole moment : CH3C1, CH2Cl2, CHCI, & CCI. b) Increasing order of dipole moment : CH3OH, CH4, CF., COCHE